Our Role in the ACT-Accelerator
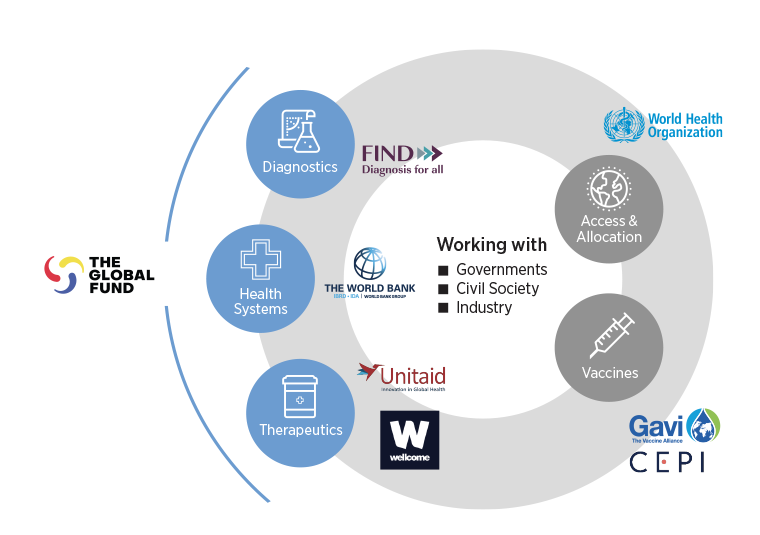
The Global Fund was a founding partner of the Access to COVID-19 Tools Accelerator (ACT-Accelerator). This global collaboration of leading public health agencies worked with governments, civil society and industry to accelerate the development and equitable distribution of tests, treatments and vaccines – and the strengthening of health systems – that the world needed to fight COVID-19.
The Global Fund was a co-leader on three of the ACT-Accelerator’s four components – the Diagnostics Pillar, Health Systems & Response Connector and the Therapeutics Pillar.
ACT-Accelerator Transition Plan – October 2022 to April 2023
The ACT-Accelerator launched a transition plan that started on the 28 October 2022. It set out how, as a partnership of global health agencies working alongside government, civil society and other partners, the ACT-Accelerator would support countries with long-term COVID-19 control. The changes to the ACT-Accelerator’s set-up and ways of working aimed to ensure countries would continue to have access to COVID-19 tools in the longer term and in case of disease surges.